Gain control of every shipment
Predict risk, prevent loss, and release products with confidence
— every lane, every partner, every time.

Predict risk, prevent loss, and release products with confidence
— every lane, every partner, every time.









.webp?width=2251&height=920&name=Pfizer_(2021).webp)
Validaide is the digital backbone for pharma transportation decisions, used by leading manufacturers to standardize lane risk, compare supplier performance, support compliant interventions, and accelerate product release.
6 of 10
top pharma using Validaide
All top 20
forwarders connected
60,000+
lanes digitized and assessed
Pharma supply chains struggle to maintain control across lanes, partners, and regions — slowing decisions, increasing operational risk, and putting compliance under constant pressure.



Connecting every partner, data point, and shipment in one ecosystem to track, monitor and release products with confidence.
Get a global view across lanes, regions, and partners to benchmark performance, prioritize improvements, and eliminate recurring risk. Make strategic decisons with confidence and support expansion into new products, lanes, and markets.
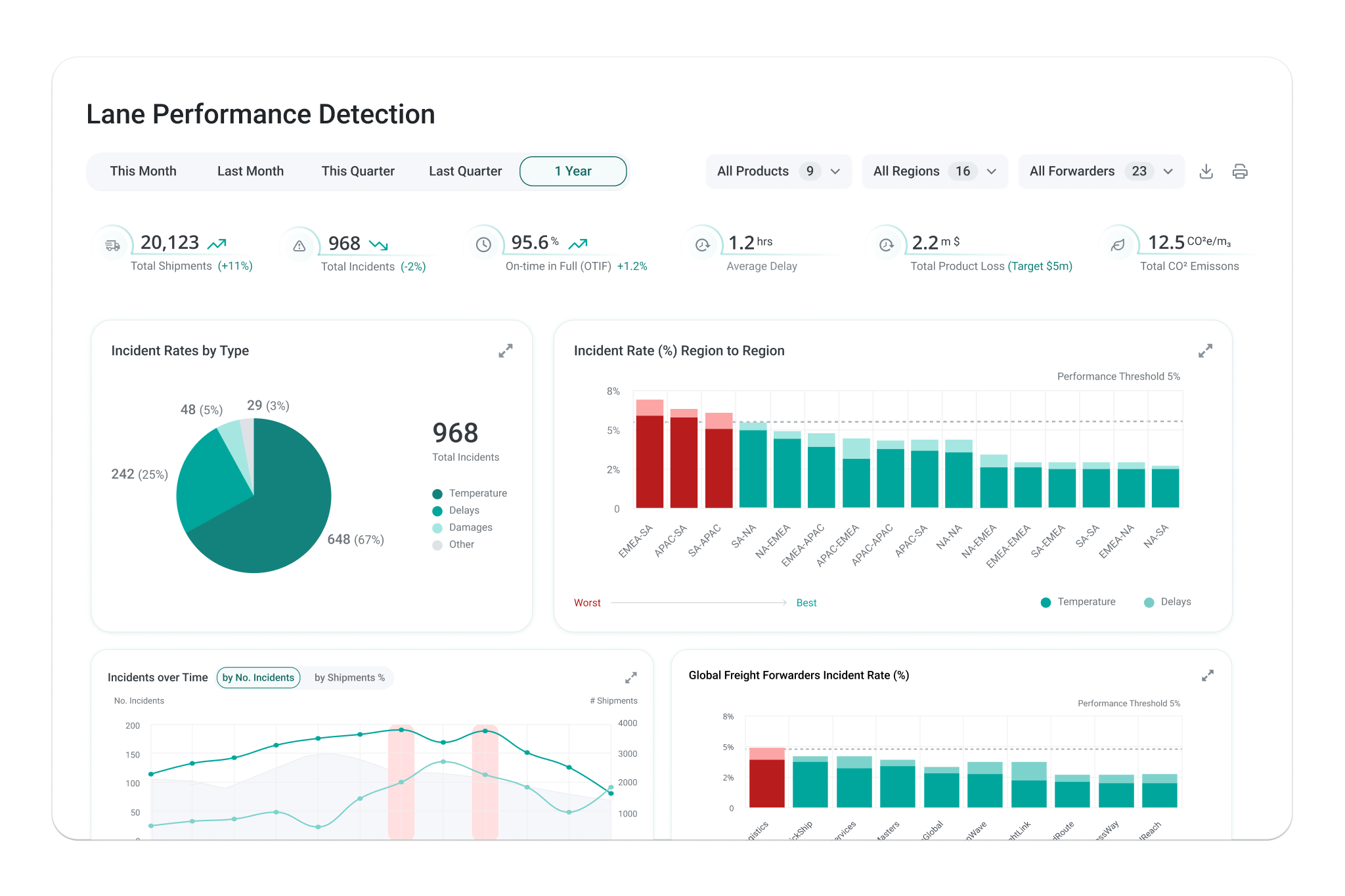
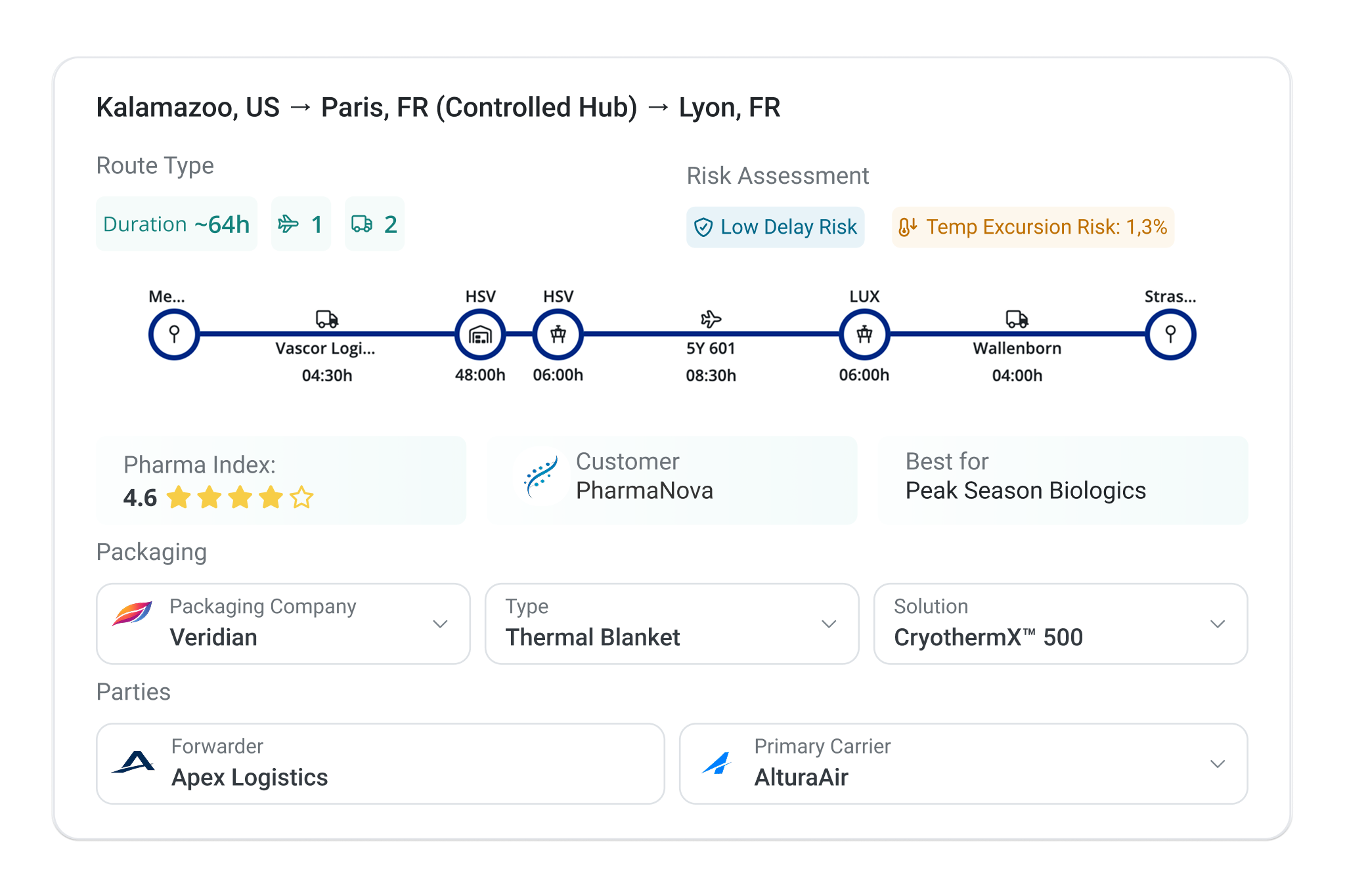
Dynamic lane risk assessments combine partner capabilities, packaging constraints, and historical data across thousands of shipments in one place. Standardize approved lanes, compare alternatives, and make changes without running test shipments.
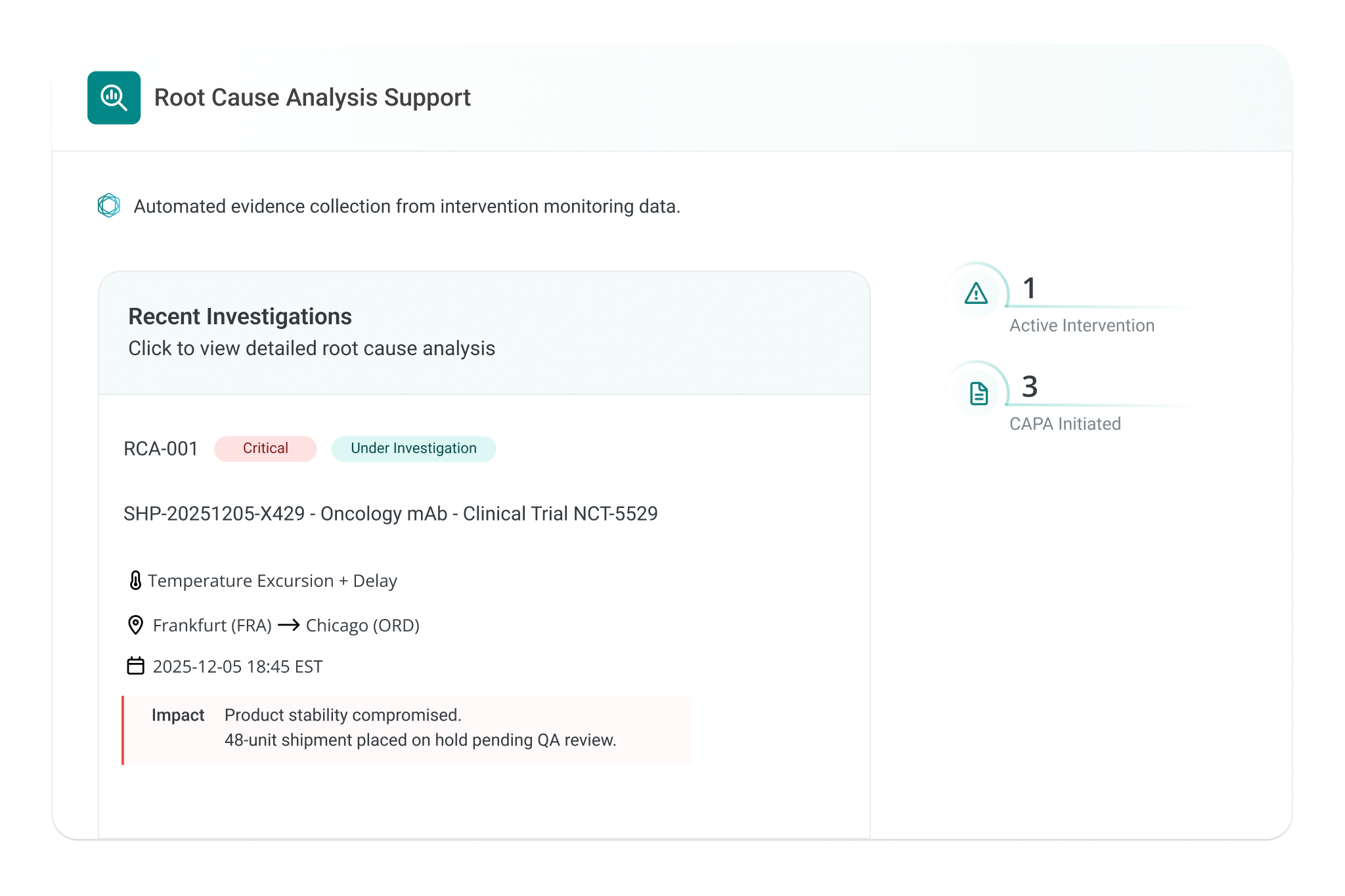
Connect real-time visibility, SOP-driven actions, and automated release into one operational flow. Detect risk early, act with clarity, and close every shipment with full traceability and compliance.

Guided onboarding, role-based training, and ongoing change support help your teams realize the full potential of Validaide in their daily work across Procurement, Ops, QA, and Logistics.
Targeted projects for lane refresh, packaging, and CO2 insights accelerate time to value and build a clear path to ROI.

Customer story
“As we enter new markets, we now have a simple, reliable way to assess risk.”
Read case study